The Truth About Vaccination
Inside India’s plan to meet the target of complete inoculation against Covid-19
 PR Ramesh and Siddharth Singh
PR Ramesh and Siddharth Singh
 PR Ramesh and Siddharth Singh
|
21 May, 2021
PR Ramesh and Siddharth Singh
|
21 May, 2021
/wp-content/uploads/2021/05/Vaccination1.jpg)
(Photo: Getty Images)
IN EARLY FEBRUARY THIS YEAR—A YEAR HAILED AS ONE OF HOPE in the war against Covid-19 just a month earlier by Prime Minister Narendra Modi—Supreme Court advocate Prashant Bhushan posted a tweet attacking the Government’s decision on an outlay of Rs 35,000 crore in the Budget for private companies, including the Hyderabad-based Bharat Biotech, for vaccine development.
Running down the outlay for ‘untested vaccines at a time when Covid was naturally dying down in India,’ Bhushan tweeted: ‘FM announces Rs 35000 cr of our money to be spent on pvt vaccine companies for untested vaccines at a time when Covid is naturally dying down in India. But this money cannot be given to poor migrant labour who lost their jobs or to farmers for MSP on their crops. Wah, FM sahiba.’ Bhushan reiterated the ‘untested vaccine’ claim a few days later with another tweet: ‘Don’t understand why people are still promoting untested vaccines in India when daily Covid cases, active cases and deaths, are down 10 percent of peak and deaths are less than 100 daily. Sero surveys show that less than 30 crore have been infected with death rates less than 1/2000.’
That wasn’t all. He claimed: ‘Delhi, Mumbai are at herd immunity.’ Bhushan is not a medical professional. The certainty with which he tweeted these opinions was not displayed even by senior doctors, learning as they were, ‘new facts and knowledge’ on a daily basis about Covid-19. Bhushan was not the only one who raised doubts about vaccines despite Modi’s January 1st address to the country in which he had announced that once expert medical advice gave the green signal, vaccine production would be amped up and a nationwide programme rolled out speedily. Addressing the key issue of vaccine hesitancy, which could prove to be a big roadblock to the programme for which his Government was preparing on a war footing, he had also exhorted the public to not fall prey to rumours about the effect of vaccines. The rumours and misinformation were being spread by those aiming to either malign the Government or individuals, or just to project themselves as persons of relevance, Modi had said, cautioning against believing such troublemakers.
A few days later, Covishield and then Covaxin were both given Emergency Use Authorisation (EUA) by the drug regulator and controversy erupted about the language used for the clearance, especially in the case of Covaxin, developed by Bharat Biotech, the Indian Council of Medical Research (ICMR) and the National Institute of Virology (NIV).
The decision by the drug regulator had been taken in the wake of one crore people being infected and another 1.5 lakh Indians losing their lives to the pandemic. Union Health Minister Harsh Vardhan was compelled to clarify on Twitter that EUA to Covaxin, which had asked for more time to submit all requisite data on clinical trials, was ‘differentially conditional—in clinical trial mode.’ Consequently, all Covaxin recipients would be tracked, monitored as if they were in a clinical trial: ‘Covaxin approval is monitored approval, with strict follow-up and rolling review.’ He emphasised: ‘For those spreading rumours, let it be known that EUA for Covaxin is differentially conditional in clinical trial mode.’

That led to more hair-splitting by self-styled experts, who even questioned the EUA since it was not a condition for approval in India under the New Drugs and Clinical Trial Rules 2019 governing nods to vaccines. It was claimed that Indians were being made subjects of clinical trials without their consent and on the pretence that Covaxin was a vaccine with all trials complete and both its safety and efficacy ensured. On social media, a storm was kicked up to claim that a ‘nationalist’ agenda of hard-selling Indian-made vaccines, with no transparency on data regarding efficacy and safety, was being adopted by the Government. This was done ignoring the facts that conditional approval meant data had to be submitted every 15 days by the makers of both Covishield and Covaxin to the drug regulator as well as the health minister’s clarification that the EUAs were given to ensure ‘vaccine security’ for a nation the size of India, for which scaling up production manifold in a short period would be imperative.
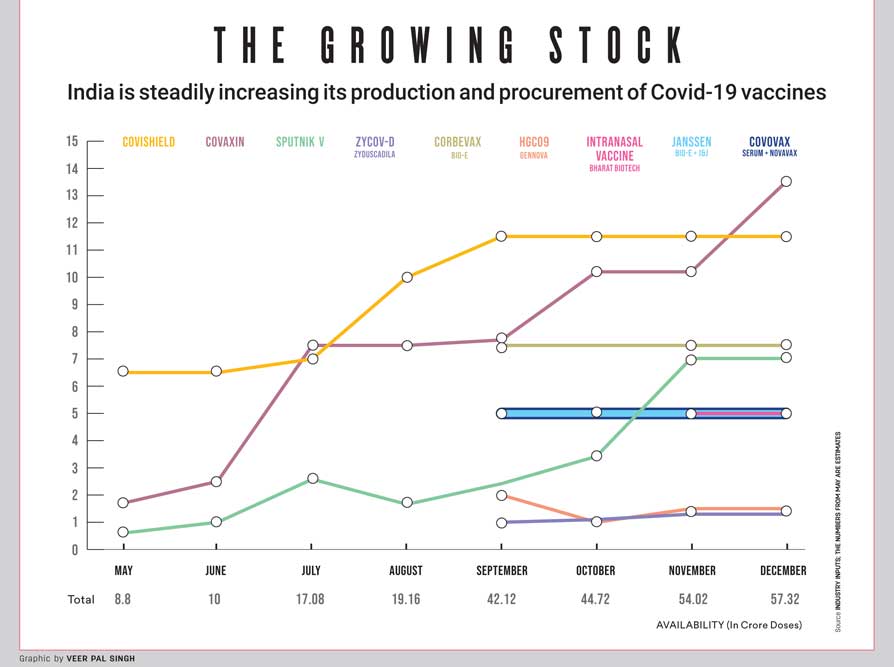
From September, more than 40 crore doses of vaccine will be available each month. It will go above 50 crore doses by November. In December, it will be 57 crore doses
Yet, the speed at which both Covishield and Covaxin arrived in response to a dangerous pandemic and were given EUA by the Government was nothing short of miraculous when compared with the mega vaccine exercises of earlier Indian governments. Take, for instance, Bharat Biotech’s Jenvac, to combat Japanese encephalitis. Covaxin, developed by the same company, was dubbed a “BJP vaccine” by Akhilesh Yadav. Yadav was the chief minister of Uttar Pradesh (UP) when Japanese encephalitis was widely prevalent in the state. Within weeks of Jenvac being launched in 2013, the virus took the lives of 15 children in Gorakhpur. Jenvac was more expensive per dose but was preferred to a cheaper Chinese vaccine for the National Immunisation Programme, presumably on account of doubts about the latter’s efficacy. A massive outlay of several thousand crore rupees for combating Japanese encephalitis notwithstanding, the state government did not take the vaccination programme for the state’s four million children to a peak level. The work on isolating an Indian strain and research on the vaccine for the disease was started by NIV as far back as 1981. It took as long as till 2013 for Jenvac to be finally launched. Bharat Biotech got in touch with NIV in 2008 to develop the indigenous vaccine as integrated into the National Immunisation Programme, more than three decades and two generations of scientists later, as the then NIV chief put it.
India accounted for more than half the deaths due to this viral infection worldwide. In this case, too, vaccine development took a long and hard road. Three decades passed from the time India recognised that it needed to develop its own vaccine and the time it was actually launched in 2015, again based on work by Dr Krishna Ella’s Bharat Biotech. That launch took place 15 years after Indian strains had been identified. Before Rotavac—the vaccine against rotavirus—India was compelled to procure vaccines from Merck and GlaxoSmithKline at more than Rs 2,000 per dose. The key driver behind the decision on an indigenous vaccine was then All India Institute of Medical Sciences (AIIMS) chief Dr MK Bhan, who was later secretary of the Department of Biotechnology. He was insistent that India launch its own vaccine against rotavirus based on the identified Indian strains and make it accessible and affordable to the country’s poor, especially since imported vaccines would become too expensive quickly. The timeline is worth noting: Phase I trials, the first of their kind for a vaccine here, started in 2003. Phase II trials were through in 2008, and Phase III trials began in 2011 and ended in 2013.
Both Jenvac and Rotavac had taken almost three times the typical timeline of 10 years for vaccine development. And there was a twist in the tale with Rotavac back then as well—once again from vaccine cynics. Prashant Bhushan was the legal representative for the petitioner who had filed a PIL in the Supreme Court to demand data transparency for better safety assessment of that vaccine.
THE CLEAR PLAN FROM THE GOVERNMENT WAS to vaccinate all health professionals and frontline workers first, and then move on to Phase II involving senior citizens and those with co-morbidities. Finally, those in the 18-45 age range were to be vaccinated in tandem with ramped-up vaccine production. The graded approach was aimed at neutralising the possibility that any trouble over the effect of the vaccines could well amplify vaccine hesitancy into an open revolt against vaccination.
Ensuring that the production of Covishield and Covaxin would be more than sufficient to keep pace with the timeline and targets outlined was a tough enough task in the world’s largest vaccination rollout programme. But it slowed down due to self-styled experts on social media and among the political opposition forcing the Government to waste precious time and resources in just countering the misinformation and disinformation campaigns.
In an effort to address that issue frontally, the Government announced a liberalised policy under which the Centre would procure 50 per cent of all vaccines produced and the other 50 per cent would be available for distribution between state governments and the private sector. The differential in prices for vaccines to be bought by the Centre and state governments was also rationalised, making it far less than the price for the private health sector. Greater elasticity was brought into effect so that state governments could take their own decisions on lockdown periods and whether to resort to them at the district level, state level or in containment zones alone. Critics of Modi, however, were up in arms once again over the price of vaccines for the Centre (Rs 150 per dose) and state governments (Rs 400 per dose) and how forcing states to resort to global tenders for vaccines at their level would enable competitive pricing to the highest bidders by vaccine manufacturers. Others contended the move would promote unhealthy price competition for vaccine purchases by state governments to the significant advantage of foreign manufacturers. Although much lip service was paid to prioritising vaccines against Covid-19 and its fast-mutating variants and associated illnesses, the key objective was to keep the pot boiling against any successful national vaccination programme taking off at the earliest.
When India prepared for vaccination, there was little inkling that a different age group would become vulnerable so soon. Preparations were based on the first wave
The developments dovetailed the usual vaccine hesitancy markers seen earlier in India. Social media and messaging platforms such as WhatsApp were at the top of the line in reiterating and amplifying unverified and incredible claims across India that the Covid vaccine would render women infertile. Rural and semi-urban India was awash with rumours, including those that maintained that the injections would trigger Covid infections in recipients. Myths spread quickly, egged on by both quacks and vaccine doubters who shored up grossly erroneous beliefs that vaccines would affect the menstrual cycle, that those who took the injection would be traced and tracked and monitored by government in all that they did, that many people had died of Covid despite two doses, making it just Government propaganda and quite ineffective against the new wave of the pandemic. Though there were such cases, they were few and far between compared to the complete recoveries from Covid-19 among those who had been vaccinated. It took sane voices from the medical profession and a concerted fight by the Government against this misinformation by affirming that the best protection against the virus was to take whatever vaccine was available and as early as possible.
By the time state governments got into the act this year to combat misconceptions pro-actively and kick off the vaccinations robustly, a lot of precious time and even more precious lives had been lost. Most state governments had wilfully bought into the widespread presumption that cases would peak in summer and ebb in winter, making for a hopeful start, both in terms of economic revival and restoring livelihoods as well as in the battle against Covid. Most had failed to scale up essential medical infrastructure and staff and stock up on drugs—and when the mutant strains attacked a younger population more rapidly and fatally, most were taken by surprise. In Ballia in UP, a group of six medical professionals who went to check on a Covid patient in a village were badly beaten. In Jamtara and Khunti in Jharkhand, the administration was forced to rope in quacks to assure the villages they served that the vaccinations were fully safe and protective against Covid. In Kashmir, it was the younger lot who had to be convinced about the injections being imperative and some took the shots with religious music playing in the background before the older citizens were convinced. In certain localities in UP, maulanas had to be roped in to send out the vaccination message forcefully. In Jharkhand, again, churches were involved in the efforts to win over cynics and in Chhattisgarh, Tribal leaders had to convince their folks that no infertility issues or chances of death would occur.
Then the vaccine crunch followed, even as the Government announced the decision to open up vaccination for all above 18.
THE US WAS THE FIRST COUNTRY TO GET OFF THE mark and race towards vaccine development for Covid-19. For all the angry pushback that it attracted, the Donald Trump administration was quick to realise that vaccination was the only long-term solution to end the pandemic. While there were hints by April 2020 that the US government was planning to help pharmaceutical companies in developing vaccines, the formal announcement came in May. ‘Operation Warp Speed’ had one key goal: production of 300 million doses of an effective vaccine by early 2021. To that end, the US initially allocated $10 billion for research and development under public-private partnership.
Later in the year, more resources were devoted to the task and, by October, the funding stood at $18 billion. Various companies were allocated funds but not all of them received funding. For example, Johnson & Johnson was given $1 billion, AstraZeneca was authorised $1.2 billion for its vaccine. Ultimately, the US stockpiled 60 million doses of the AstraZeneca vaccine but did not use it and neither did the Food and Drug Administration (FDA) approve the vaccine, due to a very small number of blood-clotting incidents reported for it. Moderna was given $1.53 billion. Novavax, Sanofi and GlaxoSmithKline were other companies that received largescale funding. These funds were made available through various laws passed by Congress in response to the pandemic. For example, the initial $10 billion funding was made through the Coronavirus Aid, Relief, and Economic Security Act, or CARES Act, that provided a multi-dimensional response to the challenge posed by the pandemic.
In Europe, the Pfizer-BioNTech project received $445 million from the German government in September 2020 to develop the vaccine using the mRNA route. By July, Pfizer had been given a $2 billion contract for 100 million doses of the vaccine.

It is worth noting that such large sums of money for research and development were feasible only in richer countries that have a well-developed research and development ecosystem. India, too, gave money to domestic companies like Bharat Biotech for research, but these amounts were minuscule compared to the gigantic sums granted in Western countries. Ultimately, India received the knowhow for making the AstraZeneca vaccine as it is one of the few countries to have facilities for largescale production of vaccines. The relative ease with which India gained access to vaccine technology was not due to munificence but for that fact of capacity and capability. It was expected to lend a hand in supplying vaccines to the world. India did that with aplomb. By May 17th, it had supplied a total of 663 lakhs doses of vaccines to 95 countries. Of this quantity, just 107.5 million doses were grants—mostly to countries in India’s neighbourhood—another 357.92 million doses were commercial sales and 198.628 million doses were under the COVAX alliance.
Ensuring vaccine production would be sufficient was tough enough. But it slowed down because time and resources had to be wasted in countering misinformation campaigns
These grants and sales were later used as political scoring points against the Modi Government even though a large fraction of these overseas supplies were important for India politically and were part of its commitments to help other countries. Had India not agreed to them, the vaccine situation would have been dire in the country. But these plain facts have never cramped the style of the opposition.
In the end, India should be able to vaccinate its population substantially, keeping in mind the target of complete vaccination by the second half of this year. Not only is the production of existing vaccines being increased rapidly but new vaccines will be added to the list. From September onwards, more than 40 crore doses will be available every month, a figure that will go above 50 crore doses by November. In December, 57 crore doses will become available. For all the doom and gloom and disruptions, India is well on its way to complete vaccination.
WHILE THE US HAD ITS OWN SET OF PROBLEMS, those did not come in the way of a successful implementation of vaccination. India, in contrast, witnessed motivated ‘vaccine politics’ right from the start of its vaccination programme. Some of these problems were evident no sooner than two vaccines, Covaxin—manufactured by Bharat Biotech—and Covishield (the AstraZeneca vaccine) made by the Serum Institute of India (SII), were given the EUA.

On January 23rd, the medical journal Lancet, in a news report, cast doubts on the trial for Covaxin and wrote: ‘Many residents recount how a vehicle with a loudspeaker had come around their neighbourhood in December, 2020. The residents allege that the announcement was made that anyone who came to the nearby People’s University private hospital could get a COVID-19 vaccine and 750 INR (£7·50). This hospital was one of the sites conducting the COVAXIN trial. Many locals have been unemployed over the past year and children have been out of school because of the pandemic, so the small sum of money was attractive enough for them to take part in the trial.’
Within no time, local politicians began dubbing Covaxin as a ‘BJP vaccine’. This was low-level politics where the target was the Bharatiya Janata Party (BJP) Government but what was also being done was to raise serious doubts about the efficacy of a vaccine, an Indian-made one, and to raise questions about the credibility of the fraternity of virologists, scientists and others. In late April, Dr Anthony Fauci, the US’ top medical expert in pandemic-related matters, stated: “This is something where we’re still gaining data on a daily basis. But the most recent data was looking at convalescent Sera of COVID-19 cases and people who received the vaccine used in India, the Covaxin. It was found to neutralise the 617 variants.” He added: “So, despite the real difficulty that we’re seeing in India, vaccination could be a very, very important antidote against this.”
In January, however, these allegations and dubious claims had a negative effect. States like Chhattisgarh—that would bear the brunt of a wildfire second wave of the pandemic barely two months later—began to see the dark side of refusing to use Covaxin. Not just politicians, top Indian scientists—including Dr Gagandeep Kang, a virologist critical of the Government—cast doubts on the vaccine’s efficacy. Although some in the medical fraternity did express some initial discomfiture about the lack of Phase III data for Covaxin, they later took Dr Fauci’s position that any available vaccine should be taken to protect oneself against Covid-19. The negative propaganda had its impact. When the first phase of vaccination was launched on January 16th, India encountered tremendous vaccine hesitancy. Even as late as March, a large fraction of the first priority group—doctors, healthcare and frontline workers—were yet to be inoculated.
When the first phase of vaccination began, it was on the basis of observing the first wave of the pandemic that had peaked in September 2020. The key takeaway at the time was that the elderly and those with co-morbidities were the most at risk. The resulting prioritisation was clear: the 300 million people to be inoculated first would be the necessary personnel needed to man medical and emergency services along with the ‘at risk’ population. The principle was sound: vaccination for those who need it and not those who demand it. Economists call this a constrained optimisation exercise: getting the most with a limited amount of resources. In the normal course, vaccinating a population of 1.3 billion people needs years of systematic mapping, training of doctors and paramedics, properly thought-out communication strategies and campaigns to limit vaccine hesitancy, and a million other details that too need to be ironed out. The war against polio in children through a universal immunisation programme has remained among the most successful such campaigns in India. It took decades for the programme to be completed. Begun in 1985, the programme continued well into the 21st century. The last cases of polio were in West Bengal and Gujarat and were reported in 2011. In March 2014, the World Health Organization (WHO) declared India a polio-free country. Even through that programme there were doubts, disagreements and criticism along its long journey.
Sometime in late February, eight districts of Maharashtra in the Vidarbha region began to show a sharp spike in Covid cases, forcing the state government to rush samples from these districts to test for new mutations of the virus. Tests in Pune earlier had shown that these were not the UK or South African variants, leading to the conclusion that these were new Indian variants. From 21 districts earlier, Covid cases shot up in 28 of the state’s 36 districts, including Amravati, Akola, Yavatmal, Buldhana and Nagpur in the Virdarbha region, and Latur, Hingoli, Parbhani and Nanded in the Marathwada region. Vidarbha was the centre of the spike, with Nagpur registering over 9,000 cases and Pune over 12,000. Amravati, Akola, Washim, Wardha and Buldhana registered the highest growth rates in cases.
The explosive increase in new Covid cases came at a time when the country had lulled itself into a sense of complacency. The first wave had been exhausting in many ways. While there was fear that a second wave would arise, as it had in many countries, people grew careless. Prime Minister Modi had spent the better part of his communication time in 2020 repeatedly messaging people that wearing masks and maintaining social distance were essential in beating the virus. By February, this message was lost. At the state-level, leaders were too busy with other matters to even pay attention to the issue of proper communication. Once again, the onus—and as later events would show, the blame—of communicating precautions fell on Modi, who became the primary political scapegoat.
The assumption behind the demand for vaccines for all was the Centre would provide. States did not bother to understand ramping-up supplies takes time
By then, however, leaders like Maharashtra Chief Minister Uddhav Thackeray and Delhi Chief Minister Arvind Kejriwal were in a state of near-panic as new cases rose exponentially. Maharashtra, for practically all of April, was caught in the grim statistics of new cases, a rising demand for medical supplies and the inability to manage the second wave. Delhi was probably worse: the mismanagement was acute and had the Union Government not stepped in with help from the armed forces, the Defence Research and Development Organisation (DRDO) and the paramilitary forces, the capital’s fate would have been much worse. But in March, leaders like Kejriwal—who had devoted attention to communicating essential measures like masking-up and social distancing only through TV and print advertisements—began imagining vaccination as an alternative to precautions. This was the first damaging step that forced a change in India’s, until then, sound vaccination strategy. In mid-March, Kejriwal said: “We should make a list of those who are not eligible for Covid-19 vaccine and make it open for the rest. It should be made a walk-in like normal vaccines. If the Centre permits vaccination for all and if we get a sufficient supply, then we have planned to vaccinate all people of Delhi within three months. I would appeal to the Centre to decentralise the process and allow states to make the decisions on vaccine matters on war footing.” He repeated his demand in a letter to Modi in early April.
This was the phase when the Centre’s vaccination policy came under intense criticism for being overly centralised. Media reports and statements from opposition-ruled state chief ministers and other opposition leaders questioned the rationality of groupwise prioritisation in vaccination. To cite another example, on March 14th, Congress leader Shaktisinh Gohil said: “The country will take 12 years in this pace of vaccination, so the states should be given more autonomy and everything should not be imposed from the Centre.” He further said that there was no dearth of vaccines in the country but the vaccination drive was slow and that was because the Centre wanted to control everything and, instead, states should be given the logistics to start the drive. With an explosive rise in the number of cases and constant carping over vaccination policy, the Centre finally gave in. It agreed to decentralise procurement of vaccines as well as relax the age-related criteria for vaccination. From May 1st, those aged between 18 and 44 would be allowed to receive the vaccine. The registration for those who were 18 and above would start on April 28th.
May 1st came and passed. After a fortnight of the liberalised vaccination drive, only 40 lakh-odd individuals in the 18-44 age group could be given their first shot. The reason was not far to seek: state governments had brought such pressure to bear on the Centre in the name of ‘federalism’ in March that the Union Government simply gave in, without states making adequate preparations for the round from May 1st. The assumption behind the demand for vaccines for all was that states would demand and the Centre would provide. State governments did not bother to check or even understand that ramping-up supplies takes time. The bravado did not stop there. Maharashtra, Tamil Nadu and some other states announced that they would float global tenders for the supply of vaccines to their states. The results were along predictable lines.
The Brihanmumbai Municipal Corporation (BMC), among the richest local bodies in India, had floated a global tender for the supply of 10 million vaccine doses. The tender was issued on May 12th and the deadline was May 18th. Not a single bid was received by the BMC. The bid was open to all major global vaccine providers, such as Pfizer, Moderna and Johnson & Johnson. It is another matter that with the exception of Sputnik V, no other foreign manufactured vaccine had received regulatory approval in India. That, however, did not deter the BMC from issuing a global tender. Similarly, Tamil Nadu, a state that recently witnessed a change of government, had made noises about sourcing vaccine supplies globally. Yet, on May 12th, new Chief Minister MK Stalin tweeted: ‘I along with other Opposition Leaders, have written to Hon’ble @PMOIndia requesting him to procure vaccines centrally from all available resources to start a free universal vaccination & distribute food grains to the needy along with cash assistance to the jobless & repeal farm laws.’ Clearly, states—especially those making the most ‘federal noises’— had realised that a world with limited vaccine supplies was not the place to float global tenders and bypass the Union Government.
This was a quick reversal from the demand for decentralisation in March. It was the harsh reality of low supplies in May, prompting the closure of many centres for vaccination for the 18-44 group, that made states adopt a realistic approach.
This, however, did not stop fatalistic commentary about India being unable to vaccinate most of its population by the end of 2022, if not later.
The reality is otherwise.
ON MAY 13TH, VK PAUL, THE MEMBER OF NITI Aayog dealing with pandemic and vaccine-related issues, outlined the supply situation for the rest of the year. He said: “Two billion doses (216 crore) will be made available in the country in five months for India and for the people of India. The vaccine will be available for all as we move forward.” The timeline sketched by Paul has been met with some scepticism. But apart from the usual risks in supply chains and manufacturing, there is little reason to disbelieve that higher vaccine availability will be a reality in the coming months.
In its affidavit in the apex court the Government said capacity expansion is already on. It is expected that SII will expand output to 15 crore doses per month and Bharat Biotech to 11 crore doses per month. In three months—May, June and July—33.7 crore doses will become available of which the Centre’s share will be 17.68 crore and the remaining doses will be available to states. By July, the combined supply from SII and Bharat Biotech will go up to 16.7 crore.
The reality is that Covid-19 is a virus about which much still remains unknown. Different countries have experienced different waves affecting different sub-populations. When India made preparations for vaccination, there was little inkling that a different age group would become vulnerable to the extreme in a very short span of time. The country’s preparations were based on what it saw in the first wave.
On March 17th, at a Covid review meeting with the chief ministers of all states, Prime Minister Modi had warned of a second wave of infections and had urged states to ramp-up medical infrastructure, drugs and other essentials in preparation for a war against the virus.
With that head start, and a whole year of the first wave, states had weeks to prepare themselves for the fight. Pandemics, hurricanes and droughts are acts of nature. But health infrastructure, supply and stocking of affordable drugs and medical essentials, setting up a working system for food rations to the needy and livelihood compensations for others, are not.
These are what elected governments should do—especially at the local and regional levels—customising their response to dire public needs in these times.

/wp-content/uploads/2025/06/Cover-OpenMinds2025.jpg)












More Columns
Indian Companies Have a Ransomware Vulnerability Open
Liverpool star Diogo Jota dies in car crash days after wedding Open
'Gaza: Doctors Under Attack' lifts the veil on crimes against humanity Ullekh NP